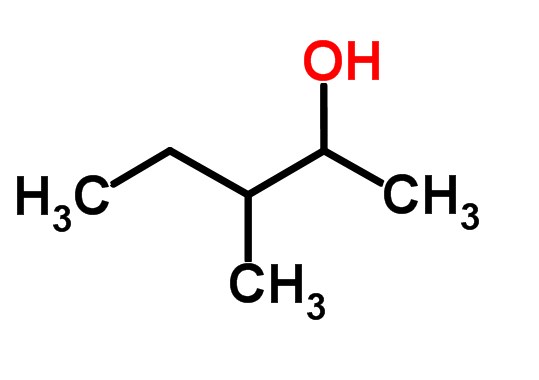
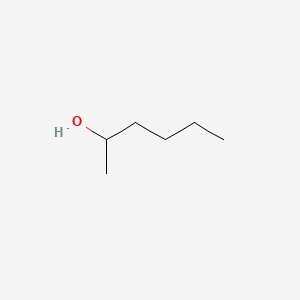
What unsaturated carbonyl compound is formed by dehydration of the below beta-hydroxy carbonyl compound? | Homework.Study.com

Separation and Purification of Methyl Isobutyl Ketone from Acetone + Isopropanol + Water + Methyl Isobutyl Ketone + Methyl Isobutyl Carbinol + Diisobutyl Ketone Mixture | ACS Omega
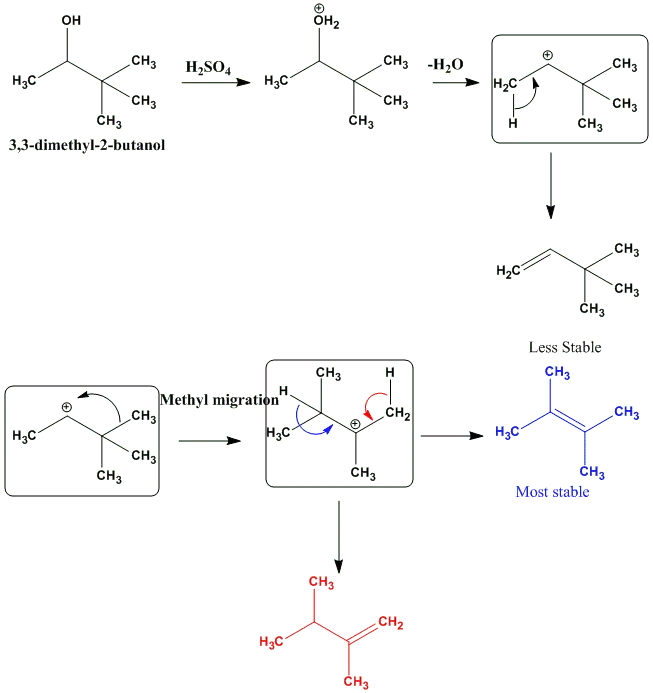
OneClass: Acid-catalyzed dehydration of alcohol 3,3-demethyl-2-butanol, togive three isomeric alkenes...

SOLVED: Spccify both the alcohol starting ' matcrial and the reagents you would use in cach stcp in a synthesis of the compound shown: If the synthesis requires only two steps enter

Dehydration of Pinacolyl Alcohol - Dehydration of Pinacolyl Alcohol Introduction H2SO4 H2SO4 C6H14O C6H12 C6H12 Figure 1. Chemical Equation for the | Course Hero

PDF) Efficient Catalytic Dehydration of High-Concentration 1-Butanol with Zn-Mn-Co Modified γ-Al2O3 in Jet Fuel Production

An ether, (A) having molecular formula, C(6)H(14)O, when treated with excess of HI produced two alkyl iodides which on hydrolysis yield compounds (B) and (C). Oxidation of (B) gives an acid (D),
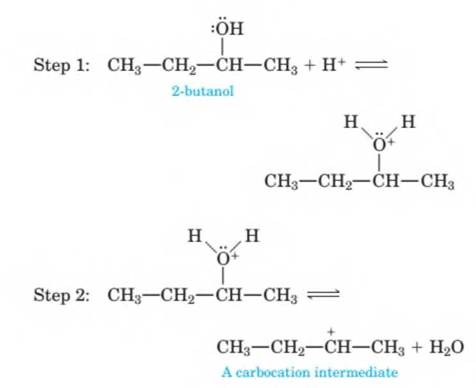
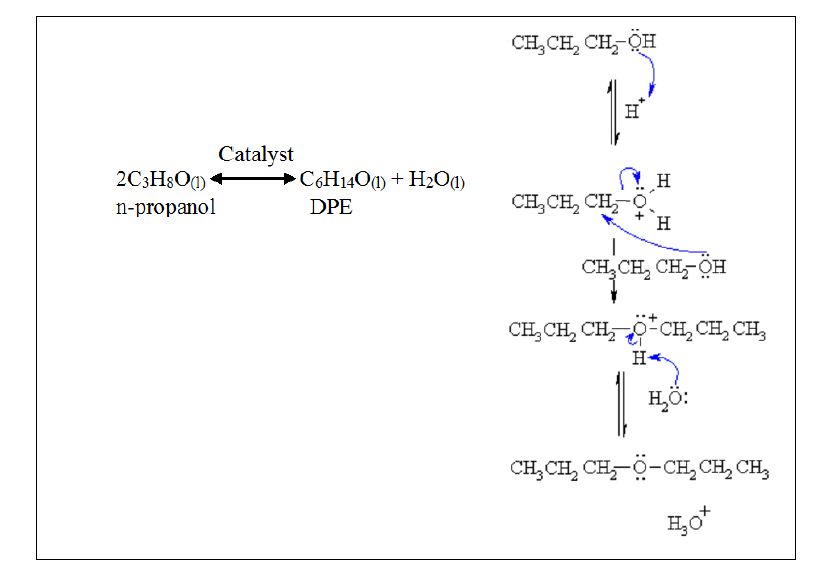
14-71 The mechanism of the acid-catalyzed dehydration of an alcohol to an alkene is the reverse of the acid- catalyzed hydration of an alkene. The dehydration mechanism occurs by the following three

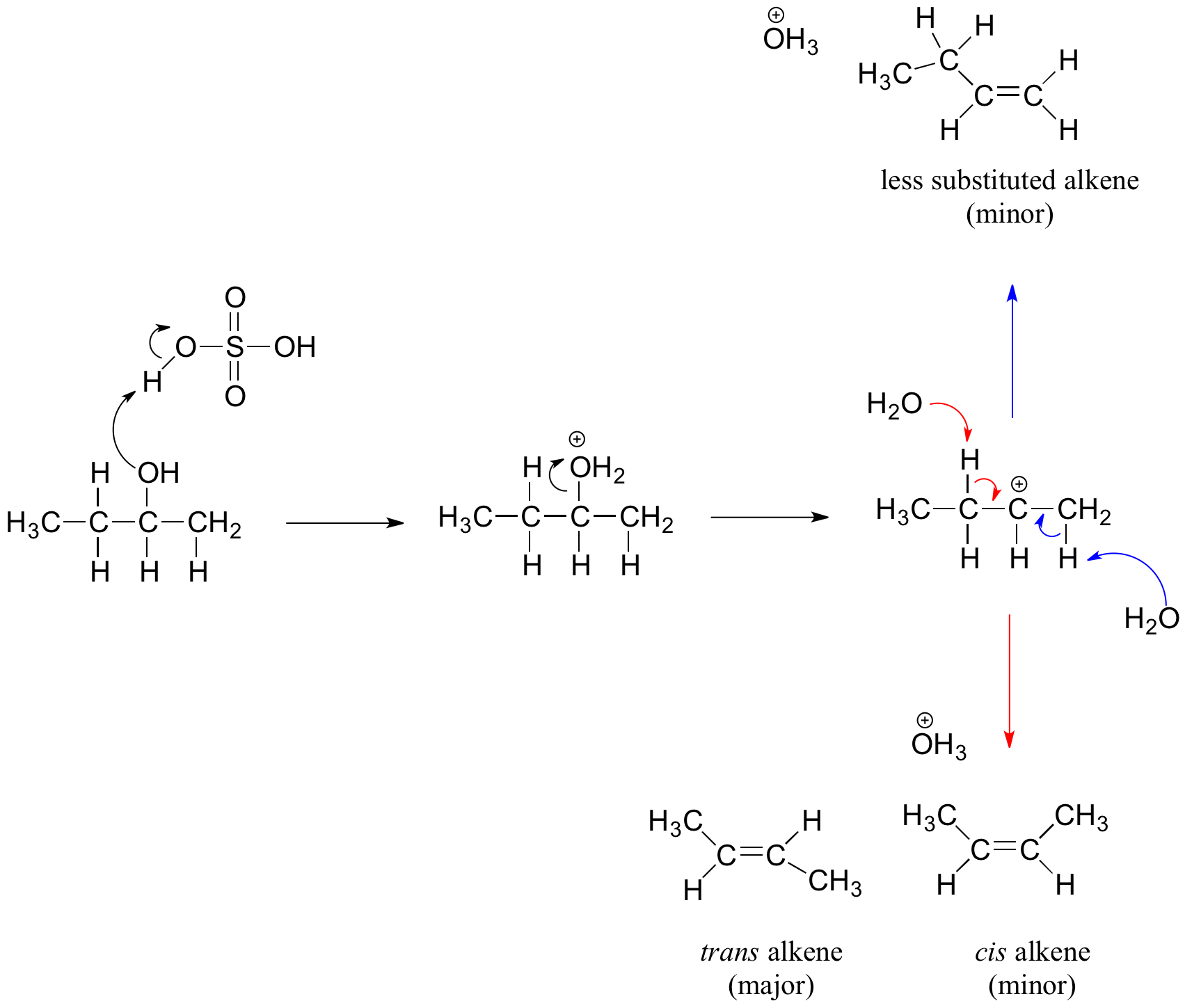
![PDF] Combinatorics of symmetric plabic graphs | Semantic Scholar PDF] Combinatorics of symmetric plabic graphs | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39f2f4dfdfdabbae5b432f4b71f2fd20dce03998/8-Figure4-1.png)