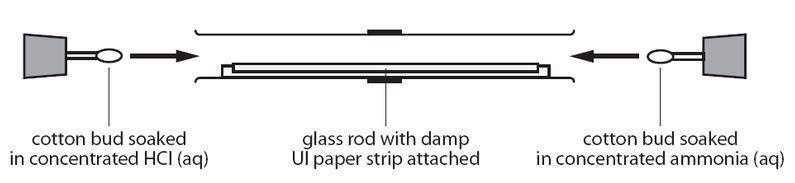
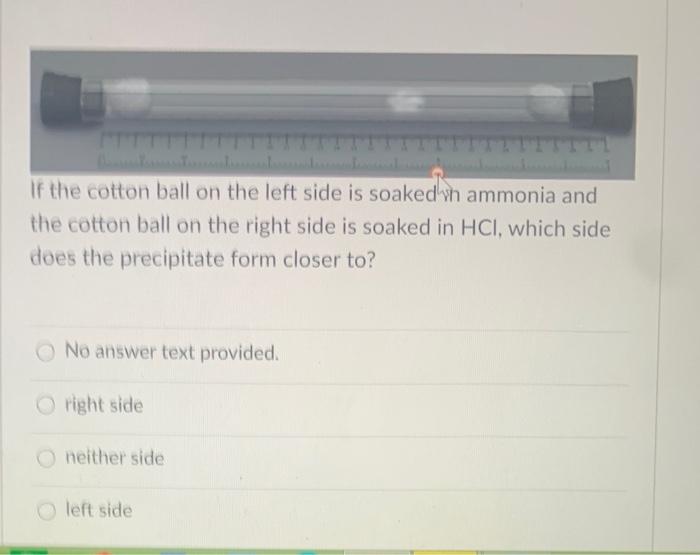
plug of cotton wool are soaked in concentrated ammonia solution and the other soaked in concentrated hydrochloric acid solution are - Chemistry - States of Matter - 16421129 | Meritnation.com
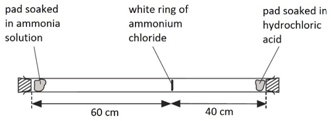
1:03 understand how the results of experiments involving the dilution of coloured solutions and diffusion of gases can be explained - TutorMyself Chemistry

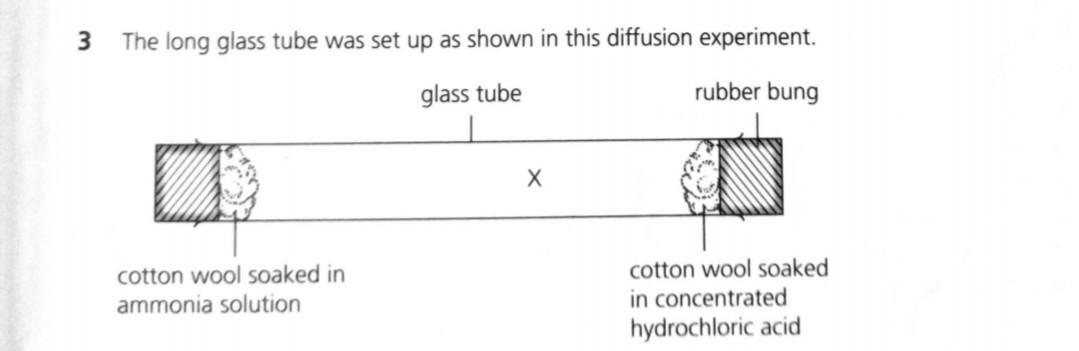
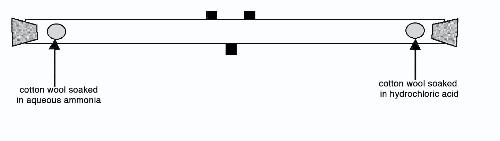
SOLVED: A piece of cotton wool soaked in Ammonia solution is placed at one end of a glass tube and at the same time another piece of cotton wool soaked in concentrated
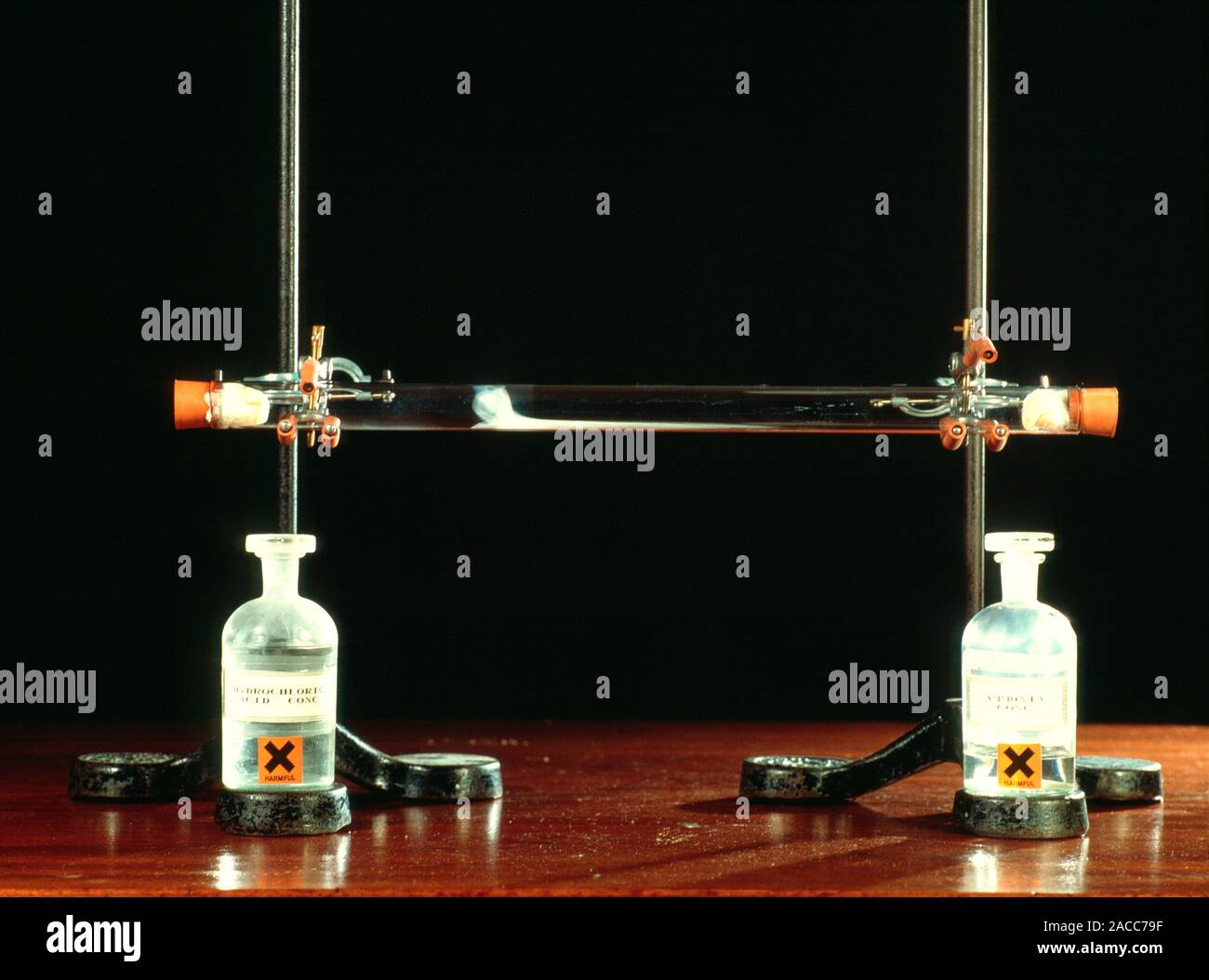
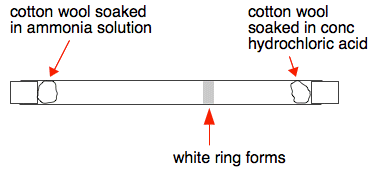
Ammonium chloride formation. Cotton wool balls have been soaked in hydrochloric acid (HCl, left) and ammonia (NH3, right). They have been placed at ei Stock Photo - Alamy
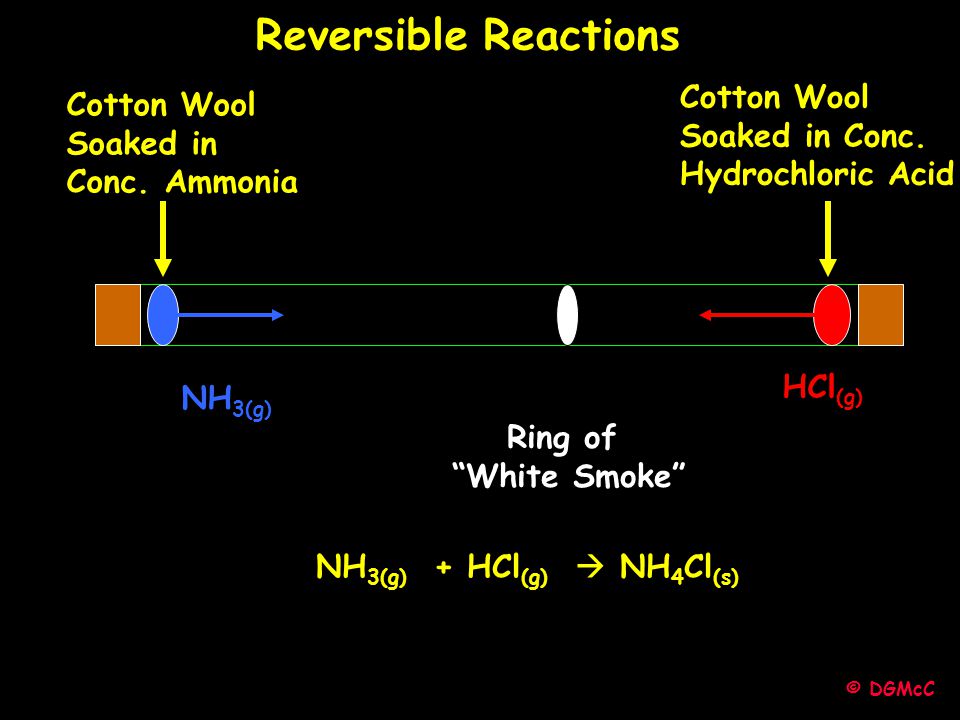
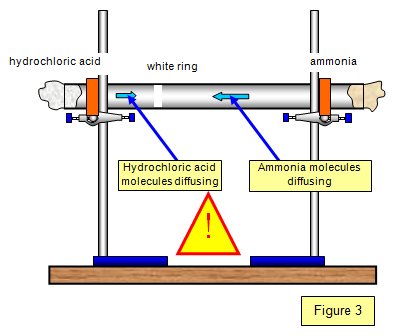
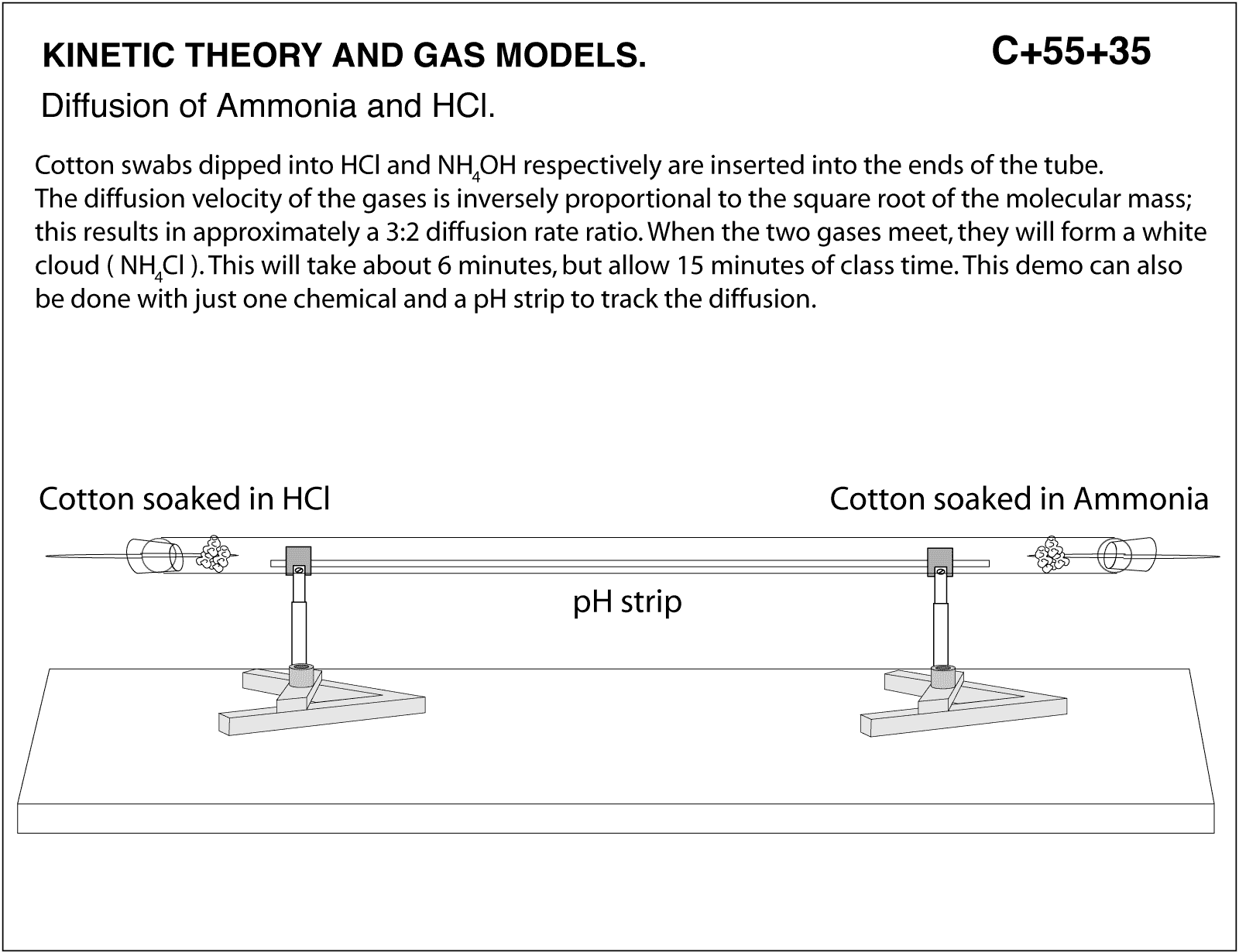
DGMcC Magee Lecture. © DGMcC Cotton Wool Soaked in Conc. Ammonia Cotton Wool Soaked in Conc. Hydrochloric Acid NH 3(g) HCl (g) Ring of “White Smoke” - ppt download

Cambridge IGCSE Chemistry Coursebook (fourth edition) by Cambridge University Press Education - Issuu

States of Matter: The Kinetic Theory of Matter (Freezing, Condensation, Diffusion, Effusion and Sublimation) - Chemistry Hive